Strategic and regulatory aspects for combination products
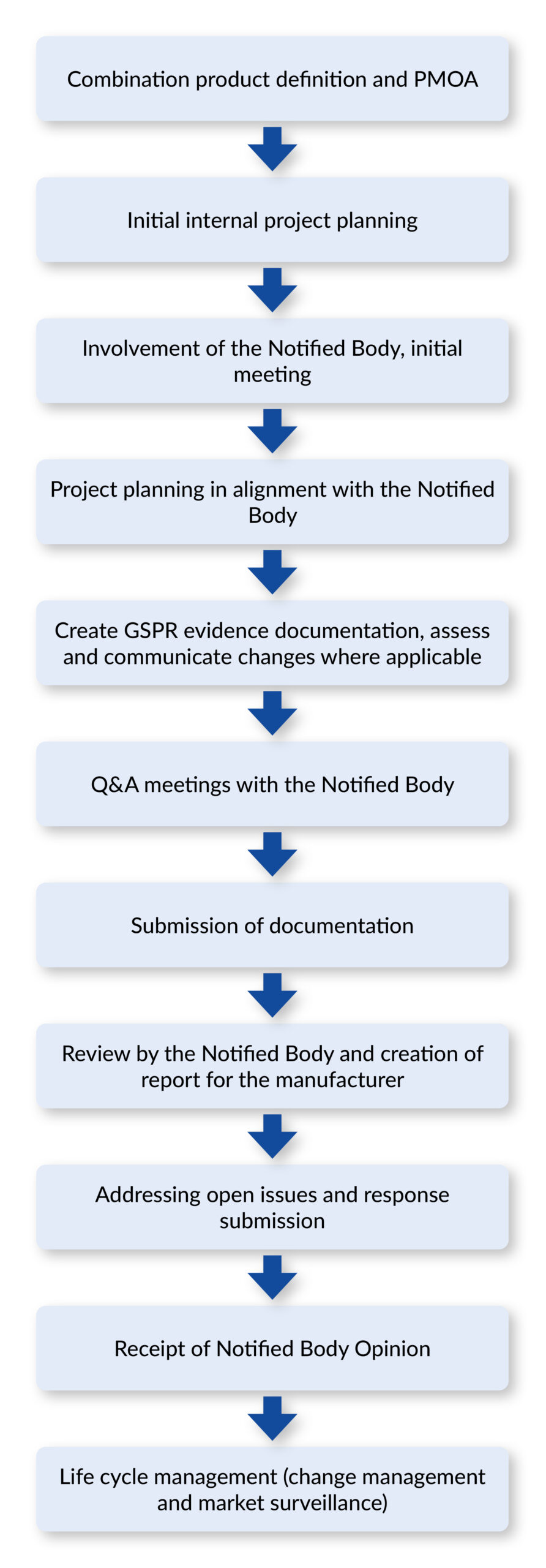
The path to your Notified Body Opinion
Marketing authorization holders of combination products are subject to requirements regarding documentation and the applicable marketing authorization procedure for the overall product, depending on its regulatory classification.
In the European Union, combination products are classified based on the main mode of action of the product. If the products main mode of action is pharmacological, immunological, or metabolic, it is subject to the medicinal products law (see also European Medicines Agency, EMA) and must be submitted to the authority as part of a marketing authorization application.
For the medical device part, it must be demonstrated that the essential safety and performance requirements of the Medical Devices Regulation are met, which is usually confirmed by an assessment by a Notified Body and by the Notified Body Opinion in accordance with the Medical Devices Regulation (EU) 2017/745 (MDR) Article 117.
What is a Notified Body Opinion (NBOp)?
According to Article 1(9) of the MDR, the involvement of a Notified Body is required for the assessment of the medical device part of integral combination products where the main effect is attributable to the medicinal product. The applicant must demonstrate that the medical device part meets the general safety and performance requirements of the MDR (EU) 2017/745 as set out in Annex I. The evidence and explanatory documentation are assessed by a Notified Body with regard to conformity. The resulting assessment report, the Notified Body Opinion, then becomes part of the marketing authorization dossier (Module 3.2.R), which is submitted to the national competent authorities (NCAs) for medicinal products in Europe or the EMA.
In addition to well-founded and coherent documentation, the prerequisites for successfully obtaining a Notified Body Opinion include, in particular, structured internal cooperation and continuous and transparent communication with the Notified Body.
An internal process for obtaining a Notified Body Opinion helps to implement the procedure in a clear, efficient, and regulatory-compliant manner.
Ready for a reliable Notified Body Opinion?
Let's work together to ensure that your combination product is optimally positioned from a regulatory perspective – from PMOA assessment to structured NBOp documentation.
How does one obtain a Notified Body Opinion?
If the PMOA analysis confirms that the main effect is based on the medicinal product, a Notified Body Opinion must be included with the marketing authorization application (MAA) for the medical device.
What steps does the manufacturer have to take?
Before starting the process of obtaining the Notified Body Opinion, it is worth taking a look at the fundamental strategic issues. This includes, first of all, assessing what development period is realistically required for the combination product. It is equally important to consider when the product should be submitted for approval - i.e., when the marketing authorization application, including the Notified Body Opinion, should be submitted to the authority.
At the same time, the framework conditions of the Notified Body must be clarified:
- How does the application process work?
- What documents does the Notified Body require at the outset?
- And what initial information must be already provided with the application form?
Another key point concerns the evaluation of possible review times. Notified bodies work with defined processing time windows that are dependent on workload. These must be integrated into internal project planning. Only when submission deadlines, development milestones, and review capacities are clearly coordinated can the manufacturer ensure that the entire approval process runs without delays.
An often underestimated point is the question regarding variants of a combination product. Clarifying at an early stage whether these can be covered by joint supporting documentation and thus in a single Notified Body Opinion is crucial for effort, costs, and subsequent change processes.
The decisive factor here is the extent to which variants affect the underlying documentation; in particular, whether and how the risk profile changes, which general safety and performance requirements (GSPR) are affected differently, and at what point adjustments are required not only to the Notified Body Opinion but also to the marketing authorization dossier. In addition, it must be examined which changes to the medicinal product could potentially affect the medical device component and thus possibly necessitate an update or a new Notified Body Opinion.
Early strategic planning, ideally in close consultation with the Notified Body, can help to reduce time and costs and significantly improve the feasibility of future changes.
Involvement of the Notified Body
It is advisable to seek dialogue with the Notified Body at an early stage in order to clarify any open questions, expectations, and requirements in advance. This enables a tailored regulatory strategy and reduces the risk of unexpected queries or delays during the assessment and approval phase.
A good start is to make the interface between the drug and the medical device transparent in the first meeting and explain the therapeutic function of the product. This helps the reviewer at the Notified Body to understand the product well and support you accordingly.
The following topics, for example, are useful to cover in an initial meeting with the Notified Body:
- Clarification of classification and confirmation of PMOA assessment
- Determination of the scope of the assessment and the documents to be submitted
- Presentation of the expected project timeline
- Preliminary development plan
- Coordination regarding the availability of the Notified Body and internal project processes
Only when these aspects have been clarified and confirmed is the basis created for drawing up a structured project plan for the development and documentation of the overall product.
Smart document structuring
Once the scope and requirements have been defined together with the Notified Body, work can begin on preparing the GSPR documentation for the medical device. In addition to the tabular presentation of the general safety and performance requirements in accordance with Annex I of the MDR and the associated evidence, explanatory documentation is required, which can be based on the structure of Annex II of the MDR.
In practice, it has proven useful to coordinate with the Notified Body on how the individual annexes are named and structured and in what form they should later be referenced in the Notified Body Opinion. This facilitates internal work and prevents misunderstandings.
It is equally important to strike the right balance in terms of the level of detail. It is crucial to focus on the content that is relevant to the Notified Body and to provide logically structured, easily understandable documentation.
It is also necessary to define how the documentation will be linked to the annexes. Here, too, a uniform structure, clear references, and intuitive navigation make it much easier for the Notified Body to find its way around quickly, thereby speeding up the entire assessment process.
During development, further Q&A rounds should be held with the Notified Body to discuss open issues or project delays. It is advisable to document the exchange with the Notified Body in a comprehensible manner and to make the minutes of the discussions available to those involved, as well as to file them in the project documentation.
What if something changes?
All relevant changes to the design, manufacturing process, risk profile, medicinal product function, or user interface must be evaluated in accordance with the standard change control procedure within the development process. The Notified Body must be involved in these design changes.
THE milestone: Receipt of the Notified Body Opinion
Once the documentation has been completed and approved internally, it is sent to the Notified Body for formal review.
The Notified Body reviews it within the contractually agreed deadlines and prepares a report with comments and any open issues. These questions or deviations must be answered and, if necessary, supplemented with additional appendices or evidence. The documentation itself is corrected in the course of the response, but is not resubmitted in its entirety to the Notified Body.
After evaluation and clarification of the queries, the Notified Body finally confirms the safety and conformity of the medical device component in the form of the Notified Body Opinion. The complete opinion usually comprises many pages and, on request, can also be provided as a summary, which is sufficient for the approval dossier.
Life cycle management of the combination products
Within the framework of life cycle management, there are two aspects that need to be considered for combination products:
Firstly: How are post-market surveillance activities regulated for the product?
In many cases, the regulations for medicinal product primarily apply here, as the product is primarily a medicinal product in regulatory terms. Nevertheless, it should be clearly and comprehensibly documented how market surveillance and reporting obligations also cover the medical device part. It is helpful to include a description of the processes with explanations in the documentation.
Secondly: Change management
Any change to the product may have an impact on the Notified Body Opinion, which is why it must be assessed whether it needs to be reported to the Notified Body or whether internal documentation of the change is sufficient. Significant changes may require a simple update, while others may require a completely new Notified Body Opinion. The Team NB Position Paper „Team NB Position Paper on Variations to Drug-Device Combinations (Significant Changes)”, and the new “Guidelines on the details of the various categories of variation [...]” (C/2025/5045) offer manufacturers helpful guidance in this regard.
In principle, a significant change is one that affects the safety or performance of the medical device part - i.e., areas that are assessed within the framework of the GSPR.
In conclusion, it can be stated that
The Notified Body Opinion (NBOp) is an essential regulatory component for combination products (medicinal products with medical devices) in which the primary mode of action (PMOA) originates from the medicinal product and the medical device component is an integral part of the overall product. According to Article 117 of the MDR (EU) 2017/745, the applicant must demonstrate that the medical device part meets the general safety and performance requirements (GSPR) listed in Annex I. A Notified Body evaluates the documentation and then prepares the Notified Body Opinion, which is subsequently included in the marketing authorization dossier.
The preparation of documentation for the medical device requires a clear structure and consistently coordinated annexes. Regular alignment and Q&A rounds with the Notified Body contribute significantly to a smooth process.
After submission of the supporting documentation, the Notified Body carries out a formal assessment, including the processing of a potential deviation report. Once any outstanding issues have been clarified, the Notified Body Opinion is issued.
Even after approval, the Notified Body Opinion remains part of the life cycle management of the combination product – particularly in the context of post-market surveillance and change management.