Regulatory Affairs

PRRC: Responsible person in medical technology companies
Article 15 of MDR 2017/745 (EU) requires that each manufacturer designate at least one Responsible Person or PRRC (Person Responsible for Regulatory Compliance) in their company.
Responsible person: Who assumes the role?
In addition to manufacturers, EU authorized representatives and other economic operators who assume manufacturer obligations also require a responsible person in their company. Small and micro enterprises are exempt from this requirement; which may also rely on an external Responsible Person, provided that permanent and continuous access to this person is granted. The Responsible Person must prove their qualification for compliance with the relevant regulatory requirements via a university degree and at least 1 year of relevant professional experience or at least 4 years of relevant professional experience.
Tasks of the responsible person
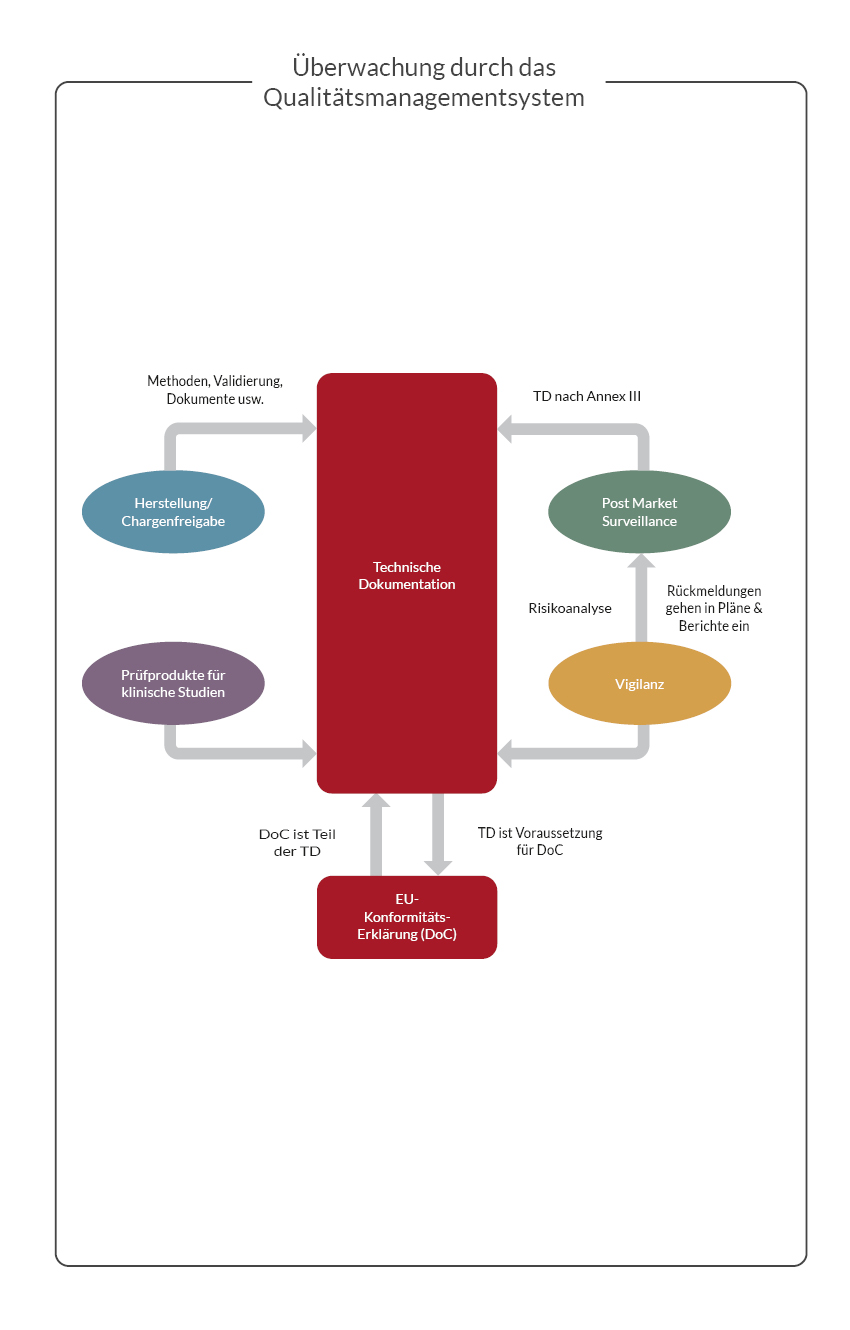
The Responsible Person is responsible for various areas. According to Article 15, these responsibilities will include, at a minimum:
- Product conformity and product release
- Preparation and updating of technical documentation and EU declaration of conformity
- Post-market surveillance
- Reporting obligations regarding serious incident reporting and field safety corrective actions
- Verification of the submission of a statement in the case of investigational devices.
The MDR also regulates that the correct performance of its responsibilities and duties must not bring any disadvantage to the responsible person.
Challenges for manufacturers and EU Representatives
While the MDR assigns the responsibility, it does not describe how the Responsible Person shall use this responsibility in practice.
Here, it is up to the manufacturer to decide whether there should be a superordinate responsible person who monitors all areas - such as technical documentation, post-market surveillance, vigilance, product conformity or the submission of a declaration for investigational devices - or whether a separate responsible person should be appointed for each area. Likewise, a decision must be made as to whether the Responsible Person will perform all of the tasks associated with assuming responsibility themselves, or whether the performance will be delegated and the Responsible Person will act primarily as a supervisory body. Once these decisions have been made, the new function of the responsible person(s), including the duties and tasks, must also be mapped accordingly in the quality management system.
In the case of an EU representative, the responsible person differs from that of a manufacturer. Here, the responsible person assumes responsibilities and tasks as a link between the EU and non-EU. The focus here is therefore less on the duties of the manufacturer and more on ensuring complete and correct documentation for the EU market, as well as carrying out the necessary registrations and communication with the manufacturer and the competent authorities.
qtec as your partner
There is no mandated way to implement a Responsible Person. However, it must be ensured that the responsible person can also fulfill this obligation. When implementing the Responsible Person, the company and the existing structures should be considered individually in order to develop a suitable strategy.
For manufacturers and EU representatives that meet the definition of small and micro enterprises, we offer in the future the possibility to access the qtec experts as an external Responsible Person to fulfill the obligations. With you, we will develop a suitable strategy and provide assistance for the implementation of the following points:
- One or more Responsible Persons?
- How can the responsible person fulfill his responsibility and what can be delegated?
- Creation of a process on how the responsibility is fulfilled
- Erstellung eines Prozesses, wie der Verantwortung nachgekommen wird
- Review of the relevant QM processes
- Review of documentation
- Creation of a QSV
Discover our "TrendReport" service
Full service for the reporting of trends in accordance with Art. 88 of the MDR. Learn more about TrendReport.
We can help!
Follow us on these pages and we will show you how we can support you. And feel free to take advantage of our free offers such as