Person Responsible - Implementation of Article 15 MDR
Since 26.05.2021, every economic operator who assumes manufacturer obligations needs a “Person Responsible” according to Regulation (EU) 2017/745 Article 15 (Person Responsible for Regulatory Compliance = PRRC). The duties of the manufacturer are described in Article 10. Article 16 describes when distributors, importers or other actors have to take over duties of the manufacturer, for example when placing products on the market in their own name – in such cases where the actual manufacturer is not present as manufacturer on the labelling.
Who needs a Responsible Person (PRRC)? - Requirements of the MDR
Every manufacturer needs at least one available PRRC within their own company. The only exceptions are small and micro enterprises, which can also appoint an external PRRC. In such cases, it must be ensured that the organization has the PRRC “permanently and continuously at their disposal”. An EU authorized representative must also have a PRRC based in the EU.
Would you like to receive further and more specific information on the topic of the MDR regulations regarding Persons Responsible? Then also read our blog post "Person Responsible according to Article 15 MDR“.
Area of responsibility of the PRRC - Challenges for manufacturers and EU authorized representatives
The PRRC is assigned a wide range of responsibilities by the MDR. These responsibilities result in a multitude of tasks, which are, however, not specified in detail by the MDR. It is possible to divide the responsibilities and the resulting tasks among several PRRCs within the organization. The additional requirements and efforts can pose enormous challenges, especially for the already mentioned small and micro enterprises, as it can be difficult to represent the PRRC correctly with the available resources.
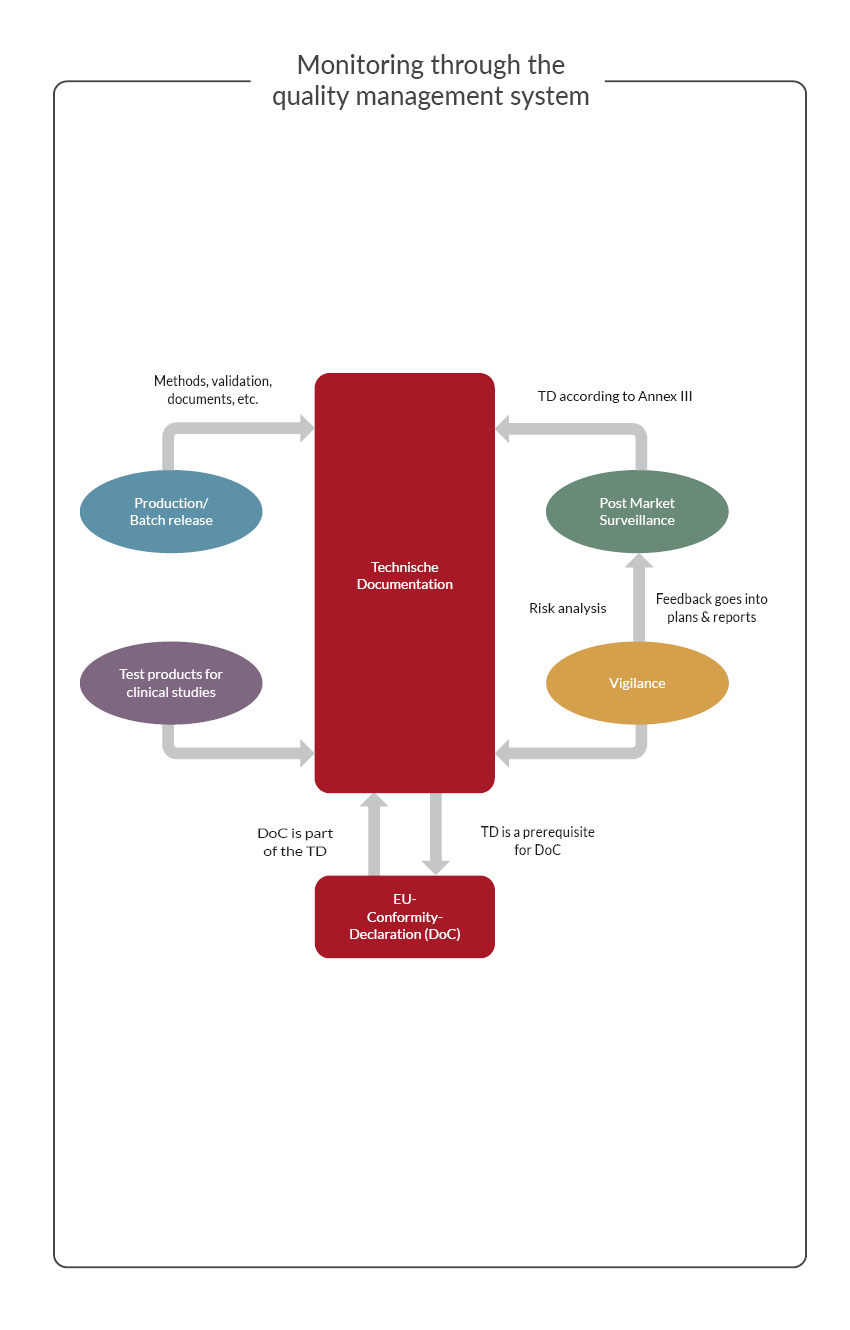
These are the relevant areas for the PRRC
Manufacturers and authorized representatives are now facing a number of challenges. What exactly is the responsibility of the PRRC and which strategies are available to meet this responsibility? Which tasks are derived from this, and which of them can be delegated? How can this be structured and established in the company? And what position is the PRRC given in the organizational chart? Are processes affected and do they need to be adapted, and if so, which ones? How to register in EUDAMED?
Download chart for PC or smartphone
Get the big picture with our chart "The PRRC at the Manufacturer."
qtec as your partner - Assumption of the function of external PRRC for small and micro enterprises by qtec
As a partner in the medical technology industry, qtec is happy to provide support in these matters and will also take over the function of an external PRRC for these companies in the future if required.
The following packages are offered for legal manufacturers:
- Product conformity and product release
- Preparation and mondernization of the technical documentation and EU declaration of conformity
- Post-market surveillance
- Reporting obligations regarding the notification of serious incidents and field safety corrective actions
- Verification of the signed statement for investigational devices
Which can be booked either individually, in combination or all together and which are preceded by an individual analysis of the needs and the appropriate model. Related tasks, such as a review of the QM system or a detailed gap analysis of the technical documentation, can also be combined with this.
For EU authorized representatives, qtec takes over the following:
- The formal review of the technical documentation and the declaration of conformity
- The review of the conformity assessment procedure
- The necessary communication with the manufacturer and the competent authorities
- Necessary registrations
Our expert knowledge for your success
We have in-depth expertise in the approval of medical devices worldwide. Ask us your questions - we will give you concrete answers and put together a project team for you on request.